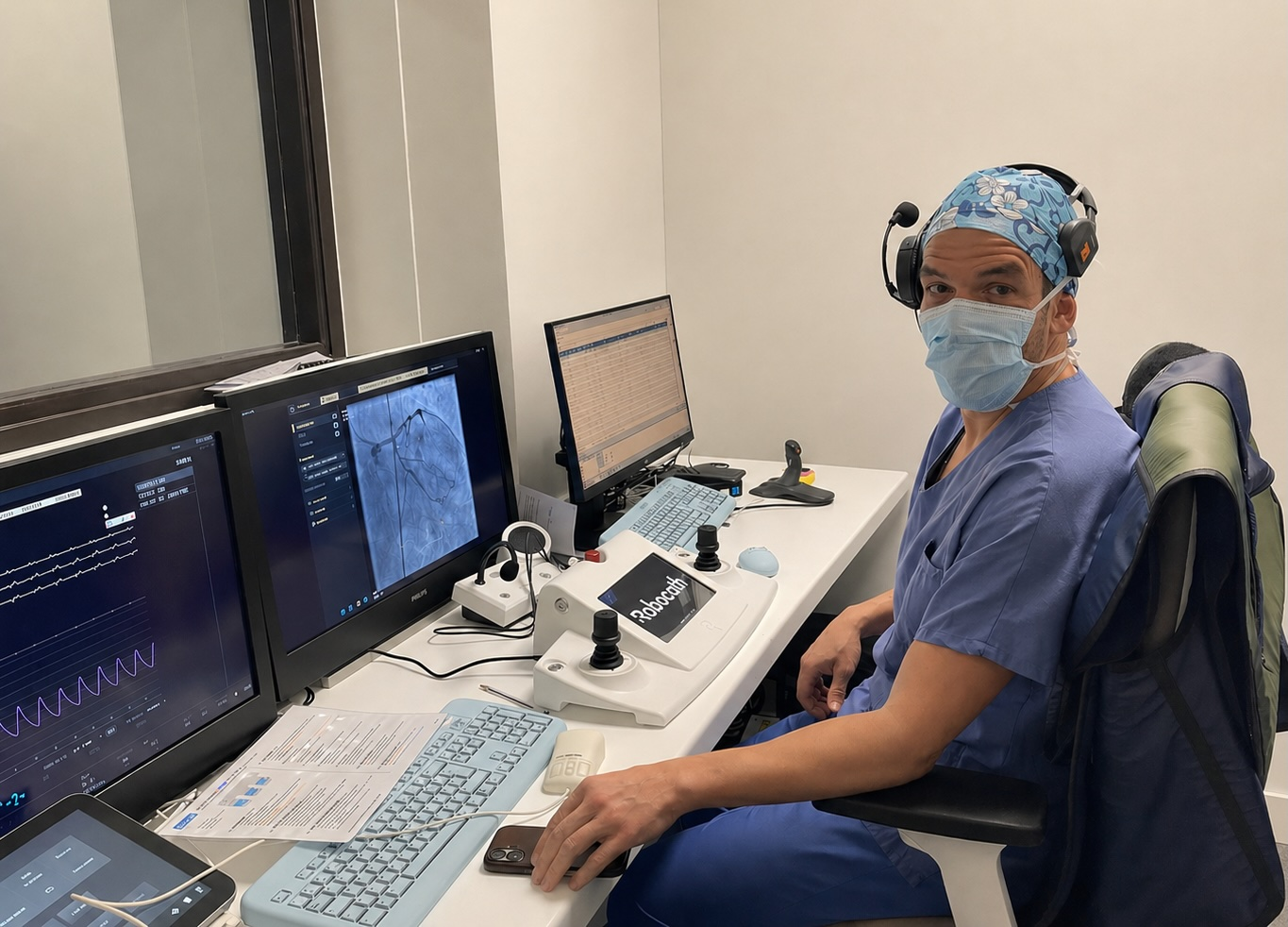
Robocath has completed patient enrollment in a first-in-human clinical study evaluating its next-generation robotic system for percutaneous coronary interventions.
Conducted at the Centre Cardiologique du Nord in Saint-Denis, France, the study is being led by Dr. Mohammed Nejjari, principal investigator, alongside Dr. Franck Digne. It is assessing the feasibility and safety of robot-assisted percutaneous coronary intervention in patients with coronary artery disease.
The trial enrolled 20 adult patients, with cases stratified by lesion complexity. Six patients had Type A/B1 lesions, seven had Type B2 lesions, and seven had Type C lesions. Robocath said no safety concerns have been reported to date in connection with use of the system, with promising technical outcomes observed across the lesion subgroups.
Robotic PCI systems are designed to give interventional cardiologists controlled device manipulation while reducing exposure to X-ray radiation during catheterisation laboratory procedures. In complex coronary cases, the ability to manage guidewires, balloons, and stents with fine movement control is central to clinical usefulness.
“Completing enrollment in this First-In-Human study is a defining moment in the field of interventional cardiology,” said Dr. Nejjari. “For the first time, we have demonstrated that robotic assistance can be safely and reliably applied to the full spectrum of coronary lesion complexity, including the most challenging anatomies.”
Robocath’s next-generation platform builds on the company’s earlier robotic technology and adds capabilities including multi-device handling. That development is significant in coronary intervention, where procedures can involve complex device sequences and demanding anatomies, particularly in Type B2 and Type C lesions.
The company’s first platform, R-One, has already been deployed in clinical settings internationally. Robocath describes its systems as robotic solutions for interventional vascular medicine, with the existing R-One+ platform designed to assist PCI procedures by reproducing physician hand movements and supporting precise navigation of balloons and stents.
Philippe Bencteux, CEO of Robocath, said completion of enrollment marked a key milestone in validating technology designed to support PCI management across different levels of complexity. He also linked the development to coronary artery disease care and the working conditions faced by healthcare professionals in catheterisation laboratories.
Full data analysis and detailed results are expected in forthcoming scientific communications. Until then, the study stands as an early clinical milestone rather than a completed performance case, but enrollment completion with no reported safety concerns to date gives Robocath a stronger basis for further validation of robotic coronary intervention.